As India moves through the early stages of the COVID-19 pandemic and puts containment procedures in place, the need for rapid and widespread testing is becoming more and more critical every day. In such a scenario, it is crucial for the public and private healthcare sectors to combine their forces and coordinate their actions. In this article, Joel and Leeba examine the need for, and efforts made towards building such partnerships in India.
Tedros Adhanom Ghebreyesus, Head, World Health Organization (WHO), told reporters earlier this week, “We have a simple message to all countries – test, test, test.” As of 23 March 2020, there are 343,421 confirmed cases of COVID-19 across the world. India has reported 415 positive cases of COVID-19 and 7 deaths. But it is likely that these numbers are so low only due to India’s low testing rates.
India has performed 18,383 tests in the 89 operational testing centres set up by the government. There are 120 sample collection centres including the testing centres. Further, 27 laboratories are in the process of operationalization. That means we now have one testing/sample collection centre per 1.12 crore people and have performed 13.73 tests per million people. With the lack of testing for numerous potential COVID-19 positive cases, it is only a matter of time before the exponential spread of the virus claims numerous casualties.
If we do not test, identify and isolate COVID-19 positive cases, we risk an outbreak as widespread as (if not more than) what has been seen in China and several European countries so far. To effectively test Indians in large numbers, solely relying on government establishments may not suffice.
The need for greater and faster testing
To understand how a virus spreads, let us say the number of cases on a given day is ’Nd’. If each person with the virus is exposed to ‘E’ number of people on a given day, and each one of those exposures has a probability ‘p’ of becoming a new infection, then the number of new cases on a given day, ‘ΔNd’ is E*p*Nd. (This does not consider recovered patients who can no longer spread the virus. Also, the actual number of cases may be more than the official counts).
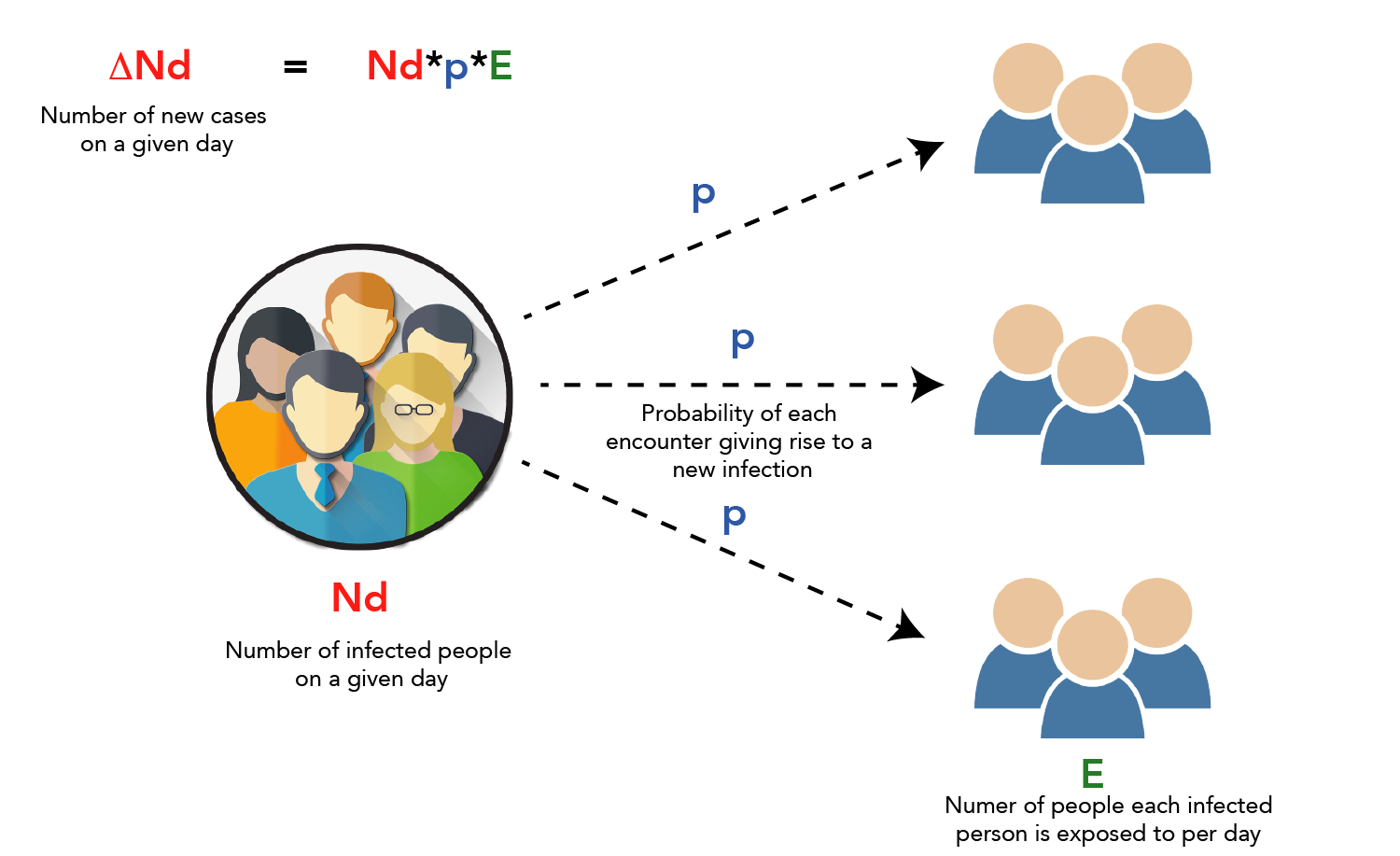
There is also the fact that Nd is a factor in itself and keeps changing as new cases are added each day. This is what allows the virus to spread so fast: as Nd gets larger and larger, the rate of growth increases as well. WHO data (COVID-19 cases outside of China) shows that the number of cases each day tends to be 1.15−1.25 times the number of cases the previous day. Therefore, while the low number of confirmed cases in India could be attributed to insufficient data, it could also be because we are at an earlier stage of the outbreak and the worst is yet to come. If we do not take the necessary steps now, it is only a matter of time before our public healthcare system comes crashing down.
One way to prevent this catastrophe is to reduce the number of new cases per day by preventing people from being exposed to infected individuals through social distancing, thereby reducing the probability of new infections.
However, social distancing can be truly effective only when we have an accurate estimate of the number of cases, especially given that a recent study found that “undocumented infections were the infection source of 79% of documented cases” in China. To prevent the spread of the disease from COVID-19 positive individuals exhibiting mild or no symptoms, we need to be testing more people.
As of now, the Indian Council of Medical Research (ICMR) is only testing those people who are showing severe symptoms of the infection or those who came in contact with people that tested positive for COVID-19. For a country of about 1.33 billion people, testing only these people might not be enough to contain the exponential spread of the virus.
Mass testing is, therefore, critical. But such an exercise requires more testing centres, infrastructure and trained professionals. This is where the government can take the private healthcare systems into confidence, bridging the gap between the public-private healthcare systems.
“We have been told that the public labs are coping quite well and are not at capacity. If the testing expands then we will need more testing capacity. Given that the private healthcare delivers most of the clinical laboratory capacity and services in the country, it is a good idea to involve them early,” Gagandeep Kang, Executive Director, Translational Health Science and Technology Institute (THSTI), Faridabad, says.
Bringing private players in
In a statement made on March 17, 2020, ICMR said that it would provide private labs with the protocols for testing. This is a positive step in bridging the gap, which needs to be implemented quickly. Testing more widely and finding negative results can reassure the people that the containment has worked. Likewise, finding positive results and quickly isolating them can also reassure people that the spread is under control.
As COVID-19 is a disease caused by a recently emerged virus, experts are also learning a lot every day. Therefore, it is challenging to manage the situation in these early days. Kang explains that while it is important to maintain clear, constant communication and engage private entities, we should also monitor testing and reporting to ensure that quality is maintained and that actions taken in response are known to public health authorities.
In the past, outbreaks like that of acute encephalitis syndrome in and around Muzaffarpur in 2019, have exposed the ailing public healthcare system in India. Taking private healthcare providers into confidence, training them where necessary and planning a cost-cutting strategy to relay the benefit to the people may lighten the burden on public healthcare system in such scenarios. Such a symbiotic association will benefit the general public greatly in the face of a healthcare emergency.
Advantages of collaborating
Public-private partnerships have demonstrated successful containment of infectious disease outbreaks in the past. In 1985, Christian Medical College (CMC), Vellore established a cell to investigate the outbreak of HIV in India. Findings of this cell led the ICMR to establish a task force to design a strategy to control the spread of HIV/AIDS. Experts from CMC guided the design of interventions, monitored its progress, and successfully contained the outbreak in collaboration with the ICMR. A more recent demonstration of the success of a public-private partnership is the containment of the Nipah outbreak in Kerala in 2018.
Although private laboratories have the capacity to test suspects during an outbreak, the costs associated with the tests is a matter of concern. An RT-PCR based test – the kind of test being used for COVID-19 – can cost about INR 5000 – 8000. Besides, there can be a surge in the prices when there is a huge demand.
“Of course, private laboratories must participate. But if private sectors are involved in the beginning, the costs of the test can be very high. So, government bodies like ICMR and Department of Biotechnology (DBT) should sit together, consider the opinions of the private entities, and frame regulations that ensure quality and cap the prices,” Abdul Ghafur, a Chennai-based infectious disease expert, says.
Hundreds of hospitals and laboratories that have been tested for quality by accreditation bodies like National Accreditation Board for Hospitals and Healthcare Providers (NABH) and National Accreditation Board for Testing and Calibration Laboratories (NABL), working around the clock from day zero, may have given us a huge leverage. But it is still not too late.
“In any outbreak, government laboratories are the first to respond. The same procedure is followed in most countries,” Ghafur says, “The ICMR has now announced that it will allow private laboratories to test for COVID-19. The government labs have done a good job so far and there has been no harm to the country by not involving private laboratories in the beginning. We have not lost any time yet and now is the right time for private entities to participate.”
The road ahead
On March 17, 2020, the government announced that it would allow private laboratories to test for COVID-19. Soon afterwards, ICMR rolled out a proforma to be filled by the private labs and laid down the guidelines for testing on March 21, 2020. The government also capped the price of the test at INR 4500 – 5000. But as of March 22, 2020, five days from the initial announcement, only six private laboratories have become functional. So while the government and private labs collaborate to catch up on getting representative data on infected individuals, it is important that we all continue to do our part by following social distancing protocols.