Many biological phenomena, like respiration, osmoregulation or nerve conduction, have a basis in physical processes like diffusion and osmosis. Definitions and diagrams may contain nuances that students may miss, especially when these are not viewed through the lens of physics. In this article, Nagarjuna G, Former Professor, Homi Bhabha Centre for Science Education, TIFR gives examples of some of these misconceptions and offers simple simulations to provide a more accurate picture of these processes.

Diffusion and osmosis are physical phenomena that happen everywhere in a living organism but they enter the discussion more explicitly while dealing with water transport in plants, gaseous exchange, osmoregulation, absorption of digested food in the intestine, and nerve transmission.
Most organs in the body and organelles in the cell are mapped to some function. If there is a structure, it has a function. So, it is roots that absorb water and minerals; lungs exchange oxygen and carbon dioxide, kidneys perform osmoregulation, the intestine absorbs digested food, axons conduct nerve impulses. This thought process is predominant in biology, which gives rise to several misconceptions.
Physical processes happen whether we like it or not, whether there is an organ or not. It is one of the features of a living cell to regulate this happenstance. Accumulating something in a location and partitioning that location from the constantly perturbing physical environment is a requirement for the origin and sustenance of life. In this process, regulating diffusion is most likely one of the first steps of living. A good grounding in thermal, chemical, and electrical phenomena is essential for understanding biological processes.
For instance, when we probe students about how we take oxygen and release carbon dioxide, they often answer that the lungs do that filtering. Lungs are incapable of making any selection of this kind since no such selection ability exists at the respiratory surface. It is pure diffusion. The students also pass the role of filtering onto RBC or haemoglobin, which are waiting in the lungs to capture oxygen and release carbon dioxide. They do not see that there is an aquatic medium in between and that the process happens entirely through diffusion. The respiratory surface is wet in all organisms, both aquatic and terrestrial. Gases must first dissolve in water before they can be used by the cells. The misconceptions related to diffusion and osmosis arise when we ignore physics while teaching biology.
Additionally, we often rely on defining terms independent of a theoretical model. Misconceptions also arise from the definitions and the terms we use. The misconceptions associated with diffusion and osmosis arise not only from the definitions of the terms “diffusion” and “osmosis” but also “solution,” “solvent,” “solute,” and popularly drawn distinctions regarding the mixtures and compounds on the one hand and physical and chemical changes on the other. Students develop mental models by learning each of these concepts independently. It is essential to situate all these concepts in a model which provides meaning to them, by invoking model-based reasoning.
Resolving misconceptions about diffusion
The definition of diffusion mentions that the process takes place from high concentration to low concentration. When we give students an example of a drop of the dye placed in one corner of a beaker filled with a solution and ask them if the dye diffuses, their answer is yes. But when we ask whether the diffusion of the dye continues after an equilibrium state reaches, most of them respond that diffusion stops when concentration is uniform across the solution. Similarly, when we present them with a beaker with a salt solution of a given concentration and ask whether there would be any diffusion, most say, “no”, since there is no concentration gradient. Students do not realize that the process is due to the dynamic and particulate nature of matter but instead remain stuck to the idea of concentration gradient, since the definition puts it so. Particles diffuse even without a concentration gradient, as long as there is kinetic energy in the substance. The dynamic character of the matter does not come to a halt after reaching equilibrium.
It is only the “net” flow in a specific direction that depends on a concentration gradient. For example, during the COVID-19 pandemic, the virus infection affected the available surface area for the diffusion of oxygen into the blood of the patients. The atmosphere has about 21% oxygen which is good enough under normal circumstances. By delivering a higher concentration of oxygen (60 – 95% depending on the severity of the patient’s case) through the nose, the lungs with the decreased surface area can be made to contain the required levels of oxygen in the blood. Thus the direction of the net flow can be regulated by a change in concentration, but the underlying process does not halt after reaching equilibrium.
One best way to appreciate the dynamic character of any substance is to know that there is a phenomenon called self-diffusion. Will there be diffusion in a substance of the same kind, say only water? Since there is no other solute, the concept of concentration gradient cannot be invoked. Water molecules move among other water molecules even at very low temperatures. Self-diffusion is known even in solids, even though the movement is relatively restricted. Experimentally such a movement is measured by using radioactive tracers (József Kónya, Noémi M. Nagy, in Nuclear and Radiochemistry, 2012).
We often do not focus on the thermodynamic nature of the solution; instead, we focus on concentration alone. Mere concentration does not help in diffusion unless the particles are dynamic. Since the dynamic character of a substance can be reduced by lowering the temperature, it is even possible to counter diffusion due to concentration gradient by thermal gradient. (See thermal diffusion https://www.thermopedia.com/content/1189/)
Since the definition is defined in terms of high and low concentration, the mental model students develop is that of crowded areas and less crowded areas. So, they attribute the cause of movement to the crowded state and not to the thermodynamic nature of the phenomena. Hence when the solution becomes uniformly crowded, i.e. isotonic, students surmise that there is no movement anymore. We often also hear from students that the particles ‘want to’ or ‘tend to’ spread out to occupy vacant spaces.
Resolving misconceptions about osmosis
Definitions also force us to think in terms of either the solute or solvent. The other most common misconception is that solvent is fluid and continuous while the solute is particulate. Definitions imply that a process is a diffusion when the solute moves from higher concentration to lower, and it is osmosis when the solvent moves from higher to lower concentration (but only when a semi-permeable membrane is present)!
When a drop of dye is placed in one corner of the beaker filled with a solvent, students think of diffusion only in terms of the dye. But the solvent particles also diffuse. Since students consider the dye as the solute, since there is no membrane, students assume that the solute alone diffuses and not the solvent. But both solute and solvent are particulate and both of them diffuse.
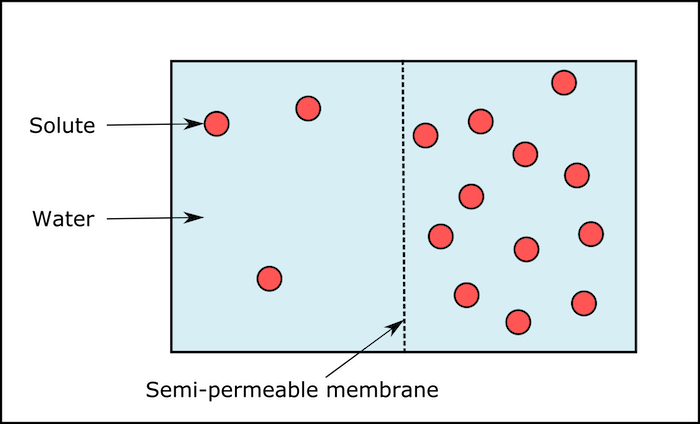
The mention of the membrane in the case of osmosis is another reason for misunderstanding. Moreover, what is the role of a semi-permeable membrane is often not discussed in detail. Neither is a correlation made between the size of the solute molecules and the size of the pores of the membrane or the polarity of the membrane surface and the polarity of the molecules. As a result, the students think of the membrane as an agent that ‘decides’ what to transmit and what not to transmit through it. They think that the membrane makes some ‘selection’ in osmosis, which allows the solvent and not the solutes. This is one reason why some students consider osmosis as an active process since an organelle is involved, and a process that stops when the cell is dead! Attributing vital characters to biological structures is a very common problem. Most biology texts avoid mathematical and physical imagination from entering into the discussion, making room for such misconceptions.
Science education research points out that misconceptions also arise because of the way diagrams are drawn. The picture shown in popular school textbooks indicates the solvent is a continuous sheet of blue colour (Fig 1), while the solute is shown as particles.
To develop a sound understanding of the dynamic particulate nature of the phenomena, one strategy that I often adopt in my workshops with science teachers is to provide a visual experience of the phenomena through computer simulations. I recommend the following simulations:
The video of the simulation shows both solute and solvent as ‘particles’. They both diffuse, and the process never stops. You can play the simulation online from the link: https://phet.colorado.edu/sims/html/diffusion/latest/diffusion_en.html
The video of the simulation shows how the solvent molecules diffuse both ways, while the membrane prevents the diffusion of solute from one side to the other, building osmotic pressure on the left side. This is shown by the movement of the membrane to the right. Please note the numbers on the left side of the screen that changes dynamically as the process takes place. You can play with the simulation online by varying the parameters from this link: Netlogo simulation showing osmosis and osmotic pressure.
In general, developing a thermodynamic view of life processes is essential for a biologist. Ignoring this, because it is physics and not biology, will not provide a sound foundation to biology and lead to several misconceptions.