The question of how stem cells can differentiate to give rise to multiple different cell lineages has fascinated biologists for years. Now, a team of researchers from the Institute of Mathematical Sciences (IMSc), Chennai and Ashoka University, Sonepat, have come up with a theoretical model that links the physical properties of the stem cell nucleus to its eventual fate.
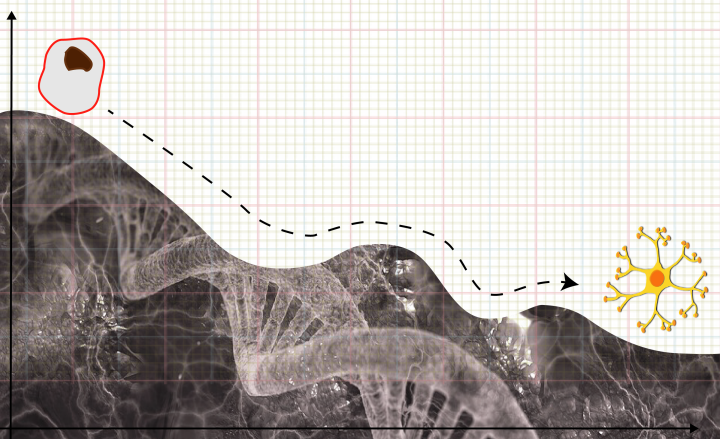
Embryonic stem cells have the fascinating ability to self-renew as well as differentiate into specialised cell types. How does a stem cell transition into, say, an immune cell? Kamal Tripathi and Gautam Menon from the Institute of Mathematical Sciences (IMSc), Chennai and Ashoka University, Sonepat respectively, have proposed a mathematical model that addresses how the packaging of genetic material, also called chromatin, plays a crucial role in deciding the fate of a cell.
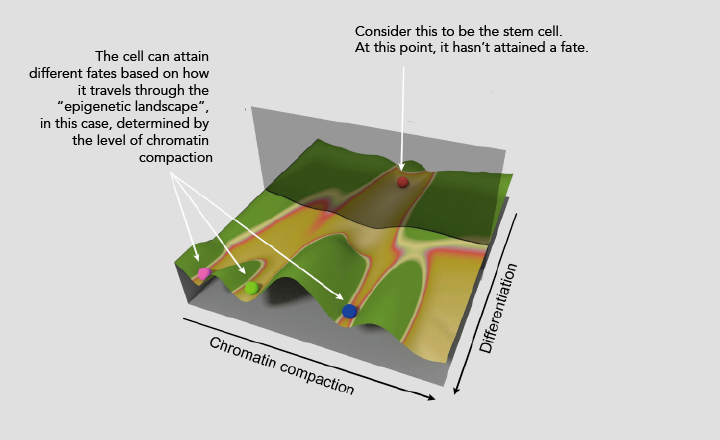
In the 1940s, Conrad Hal Waddington put forward the idea of an “epigenetic landscape” to explain how stem cells develop into specialised cells over time. Cells can control which genes are read and which are not through biochemical modification of DNA or DNA-binding proteins. This process, known as epigenetic modification, can push the cell towards different fates.
In his book ‘The Strategy of the Genes’, Waddington explains the “epigenetic landscape” with a simple, albeit poetic, analogy. He compares the stem cell to a ball that is rolling down a slope. The ball has to travel through a terrain that is riddled with ridges and valleys, which alter its path. The diverse paths arising thus mirror the many possible fates of the stem cell.
This picture of the epigenetic landscape was not directly backed by experimental observations. Questions loomed, especially those which called for greater clarity on what these “ridges” and “valleys” represent.
One answer to this might be that the “ridges” and “valleys” depict different packaging states of chromatin within the nucleus of the differentiating stem cell. These features of the epigenetic landscape are influenced by factors that bring about epigenetic modifications in stem cells. In this regard, Menon and Tripathi’s study proposes that the interplay between mechanical properties of the stem cell nucleus and the way its genetic material is packaged may sculpt the epigenetic landscape of the cell, influencing its fate.
Recent studies have shown that mechanical forces on the stem cell nucleus can regulate how genes are expressed inside the cell. Upon probing this phenomenon further, researchers found that the stem cell nucleus can behave rather peculiarly when exposed to external forces.
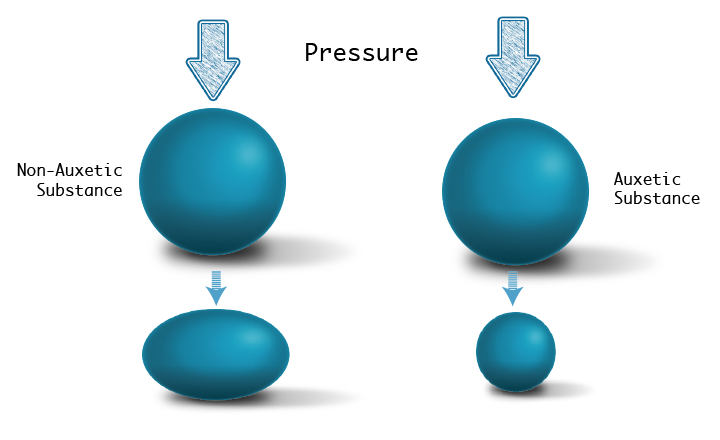
Most materials, when compressed from the top, flatten a bit. If you compress a ball of playdough with your palm, it would become slightly elliptical. Surprisingly, the nucleus of mouse stem cells that are transitioning into a differentiation primed state exhibit the opposite behaviour, known as auxeticity. Now, if the ball of playdough was auxetic, instead of flattening out when compressed, it would decrease in size, forming a smaller, denser ball of the same shape. Experiments have shown that the transitioning nucleus in mouse stem cells also decreases in size when it experiences a similar compression.
Unlike transitioning stem cells, the nucleus of a differentiated cell does not display auxetic behaviour. At the same time, the chromatin in transitioning stem cells is loosely bundled while differentiated cells have more densely packed chromatin. A previous study in the field drew a potential link between auxetic behaviour and loosely bundled chromatin in ‘transitioning’ stem cells. Menon says, “The experiments hinted that understanding chromatin compaction was central to figuring out how the stem cell nucleus responds to forces.”
Menon and Tripathi took this idea further to ask whether the level of chromatin compaction could be used to understand the stem cell’s epigenetic landscape, and hence its fate. Based on previous experimental observations in the field, the researchers identified two relevant variables to arrive at a simple theoretical model — (1) How compact is the chromatin? (2) What happens to the size of the nucleus when compressed?
The authors began by designing mathematical models to predict the mechanical properties of the stem cell nucleus. In these models, the transitioning stem cell nucleus shows auxetic behaviour when compressed, substantiating what has been observed in previous experimental studies. The model arrives at a correlation between two factors — (1) the compaction of chromatin and (2) deviation of nuclear size from an uncompressed value. This could possibly be used to predict the point when an embryonic stem cell transitions into a more specialised cell.
Tamal Das, a mechanobiologist from TIFR Centre for Interdisciplinary Sciences who was not associated with the study, refers to the auxetic behaviour of the stem cell nucleus and the increase in rigidity while it differentiates over time, as “two most important and intriguing observations in nuclear biology”. He says, “This work uniquely attempts to provide a single mechanistic explanation for both while considering the nucleus as a polymer material that can sense and respond to any stimulus applied on it.”
While this study establishes how forces experienced by the stem cell nucleus play a crucial role in regulating differentiation, it also raises new questions. For example, is it possible that similar forces may be exerted on the cell by its neighbouring cells or the substrate on which it grows? The authors also suggest that one could probe how these “ridges and valleys” in the epigenetic landscape may change over time.
This study asserts the need for a deeper investigation into the factors that allow the epigenetic landscape to be dynamic. This interplay of multiple elements that tip the stem cell towards certain fates is complex, but nonetheless, an extraordinary affair.