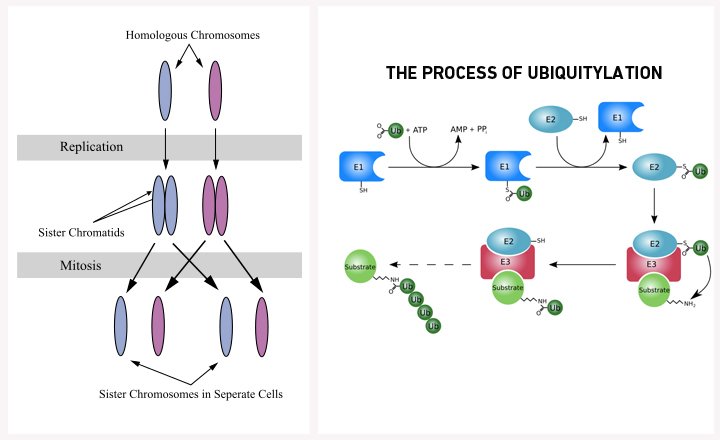
During cell division, the diploid genome doubles. The pairing, alignment and eventual separation of similar chromosomes (called sister chromatids) allow the duplicated genome to be distributed evenly between the two daughter cells. While the cellular machinery regulating sister chromatid cohesion is still being understood, a recent study led by Subbareddy Maddika at the Centre for DNA Fingerprinting and Diagnostics (CDFD), Hyderabad reports the discovery of a novel player in this process, a multi-protein ubiquitin ligase complex called CRL7SMU1.
Like other ubiquitin ligases, the CRLs (Cullin-RING-type E3 ligases) help the cell carry out ubiquitylation — the covalent attachment of ubiquitin (a small 76 amino acid protein) to a target protein or substrate. Apart from its best-known role of marking proteins for proteasomal degradation, ubiquitylation also regulates multiple cellular processes like endocytic trafficking, DNA repair, transcription and translation.
Ubiquitylation involves three steps. First, E1 enzymes ‘activate’ ubiquitin. Next, E2 enzymes conjugate with the activated ubiquitin and finally E3 ligases transfer the ubiquitin to target proteins. E3 ligases are particularly important because they determine the substrate-specificity of ubiquitylation complexes. Out of three different types of E3 ligases found in cells, RING-type E3s are the most abundant and together with Cullin proteins, form the CRL multi-protein complexes.
One of the long-standing interests of Maddika’s lab is to understand the role of ubiquitin ligases in various cellular processes. From previous studies, the group recognised the ability of proteins containing a domain called LisH to facilitate substrate recognition by E3 ligase complexes. Curious about other LisH-domain proteins in the genome, the authors performed an in silico search, identifying 28 such proteins. One of these was SMU1, a protein with previously reported roles in DNA replication and repair as well as mitotic spindle assembly.
In order to find out if SMU1 can assemble E3 ligase complexes, the researchers used recombinant SMU1 as a bait to fish out all the proteins in a cell extract that can bind to it. Out of the many candidate proteins so identified, the scientists found a scaffolding protein, an adaptor protein and an E3 ligase. “Together these components can make an E3 ligase complex,” said Varun Jayeshkumar Shah, first author of the study. Subsequent experiments proved this hypothesis right and led to the identification of CRL7SMU1, a novel multi-protein E3 ligase complex.
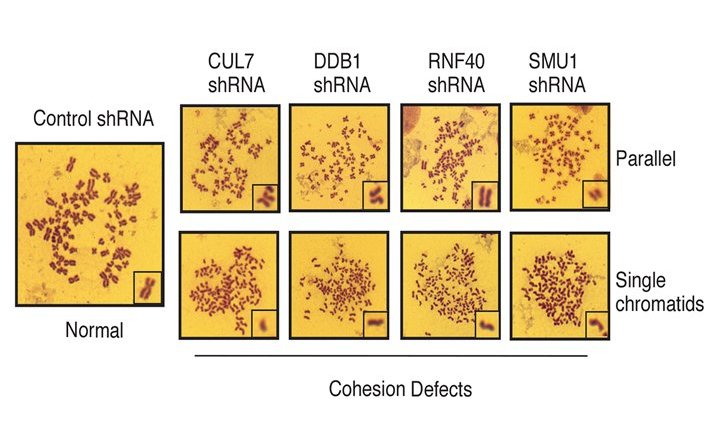
Interestingly, when the researchers knocked out components of this complex in cells, they observed mitotic defects and polyploidy, along with changed expression of a subunit of the cohesin complex, which plays a vital role in the cell division process by stably but reversibly connecting the two sister chromatids.
Elaborating on this discovery, Shah said, “Identifying the ligase and the substrate from multiple candidates in the list was tough. But, I was extremely excited to find mitotic defects in cells where the complex components were knocked-down, hinting at its biological function”.
But how exactly does CRL7SMU1 influence the cohesin complex? The researchers identified the histone, H2B as one of SMU1’s interactors. Histones allow the wrapping, packaging and compacting of DNA into the small nucleus of a cell and post-translational modifications of histones regulate gene expression. Connecting these clues, the scientists showed that CRL7SMU1 ubiquitylates H2B, thus regulating the transcription of the crucial cohesion complex subunit.
“Though the underlying mechanisms are still unclear, the role of CRL7SMU1 in sister chromatid cohesion is another interesting finding of this study,” said Ranabir Das from National Centre for Biological Sciences (NCBS), Bengaluru, who was not associated with this study.
Maddika’s lab continues to work on LisH-domain proteins and is keen on identifying other ligase complexes formed by them. Many ubiquitylation pathway components are tumour suppressors and oncogenes. Their emerging clinical significance makes it crucial to identify this diverse cellular ubiquitylation arsenal. “The number of E3 ligases has doubled since my postdoctoral days, and their diversity is only expected to increase,” concluded Maddika.
=====================
Credit for Inline images:
Left Panel: SyntaxError55 at the English language Wikipedia [GFDL or CC-BY-SA‑3.0] from Wikimedia Commons
Right Panel: Rogerdodd [GFDL or CC-BY-SA‑3.0] via Wikimedia Commons
=====================
Find this story interesting? Please comment below to let us know your views on this new discovery.