![One of Benedict Arnold's coded communications with lines of text in invisible ink [Public Domain] via Wikimedia commons](https://indiabioscience.org/imager/articles/15263/ArnoldCipherLetter_4b32b63c5c28c858e051e9d1a2a717a1.jpeg)
“Khamosh se afsana, paani se likha hota,
Na tumne kaha hota, na humne suna hota”
- Gulzar
Secrecy of messages is imperative in romance and espionage alike; self-destructible secret messages provide an added thrill. Recently, a team of scientists devised a way to use sunlight as an invisible ink to write secret messages (instead of evanescing water as penned by Gulzar!).
On a hot summer morning, Manas K Panda (Scientist at the Photonics division, CSIR-National Institute for Interdisciplinary Science and Technology (NIIST), Thiruvananthapuram) left green fibrous crystals of a newly created compound on a piece of filter paper. He forgot to observe the crystals the next day — and the next, until a chance observation 15 days later. While he remembered the crystals to be green in color on the day of preparation, now they were orange-red.
Since Panda had been synthesizing compounds that could switch fluorescence, he instinctively observed the crystals under ultraviolet (UV) light. The crystals exhibited a shift in fluorescence– emission maximum was at 535nm for the green crystals, but at 640nm and 580nm for the orange-red. Panda observed that compared to the luminescence shift, the fluorescence shift was much more remarkable.
What happened in those 15 days? Panda and his team had synthesized a chemical (which they named Compound 1) that altered fluorescent properties based on its solvent. When Compound 1 was precipitated from DMSO (dimethylsulfoxide, a polar solvent), it fluoresced in the green spectra. However, when the residual DMSO entrapped in the crystals evaporated, either rapidly on heating, or slowly on exposure to sunlight (or on leaving it at room temperature for days, as Panda had done), the crystal began to fluoresce in the red spectra. This fluorescence (or luminescence) switching property is known as solvent-dependent vapochromism.
Vapochromic compounds offer an interesting use – they can serve as invisible ink for steganography, the practice of hiding a secret message within a seemingly innocuous script. Most biological fluids (blood, urine and semen) have been used as invisible inks — interesting accounts of their use come from the period around WWII. Shining UV light (or applying a little chemistry) allows access to these messages. These properties of biological compounds have inspired chemists to synthesize sophisticated, responsive and destructible inks.
Panda and co-authors put the vapochromic feature of Compound 1 to use in this manner: they rubbed the fibrous crystals of Compound 1 on a sheet of cellulose paper and selectively exposed portions of the paper to sunlight by overlaying with a stencil, allowing the exposed areas to fluoresce differently than the unexposed region. Since the change in color of the Compound 1 on the cellulose paper was too minute to be detected by the naked eye, the code could only be read under UV light. To destroy the message, the cellulose paper could either be heated or exposed to intense sunlight, essentially evaporating entrapped DMSO from the portions masked by the stencil, making the original message illegible.
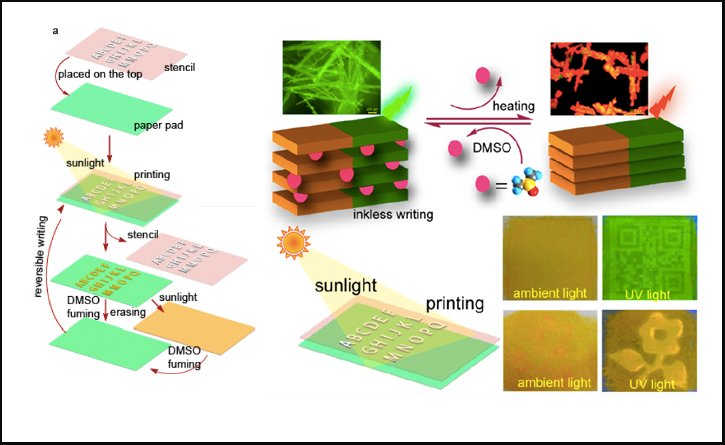
Many scientific groups have been working on fluorescent compounds (with metal-organic frameworks (MOFs) as template), which have garnered attention as potential tools for confidential data communication – however, compared to a sheet of cellulose paper they are expensive. Panda suggests two other advantages over MOFs: “There is no threat of heavy metal exposure, and cellulose papers can be re-used multiple times.” With the help of a few collaborators, his team plans to pursue biological research on the cytotoxicity of Compound 1.
Could this method be reversible? Panda says, “We have tested the reversibility of sunlight-pen writing for 3 – 4 cycles. However, it greatly depends on the life of the cellulose paper.” He also points out that an advantage of his method is that on a laboratory scale, the purification of the compound is easy, since it easily precipitates out. “The issue of scalability needs to be taken up,” he adds.
Bipin Kumar Gupta, scientist at the CSIR-National Physical Laboratory (not associated with the study) finds this approach unique and creative. However, he is concerned about the stability of the compound and did not find the quality of printing to be very crisp and sharp. “Confidential data communication requires rapid destruction, however exposure to sunlight would take the message about 4 hours to destroy,” he adds.
Did you enjoy this article? Let us know in the comments below.